[China Aluminum Industry Network] In order to expand the application of aluminum and aluminum alloys, a new combination of aluminum leaching, alkaline electroless nickel plating, acid electroless nickel plating, and chemical imitation gold plating has been developed. Aluminum alloy chemical dipping gold imitation gold technology, discussed the main components and process conditions on the quality of imitation gold coating, determine the process conditions are as follows: SnSO48 ~ 10g / L, CuSO41.2 ~ 1.5g / L, coordination agent (tartaric acid or Citric acid) 10~15g/L, H2SO410~20mL/L, XT-08B stabilizer 10~12mL/L, hydrofluoric acid 40~50mL/L, ammonium fluoride 1~2g/L, temperature 15~35°C , time 10~15min. The resulting gold-plated coating is elegant and pure in color, has good bonding force, is simple in process operation, and has less pollution to the environment, and the corrosion resistance can be comparable to the electroplated imitation gold layer, and has a good application prospect.
Key words: Aluminium; Alloy; Chemical immersion plating; Gold-like imitation; Corrosion resistance. CLC: TQ153.2 Document code: A
Article ID: 1004–227X(2012)03–0012–03
1. Preface Gold is deeply loved by people for its luxurious, solemn and elegant decorative effects. With gold decoration, the cost is too high and ordinary people are discouraged. The imitation gold plating obtained by the electroplating method has a color of 18K to 22K gold, and the cost is greatly reduced, which can meet the needs of ordinary people for golden decoration. At present, the imitation gold plating is mainly realized by electroplating method, and its application process is cyanide-plating imitation gold or pyrophosphate plating imitation gold [1-2].
Among them, cyanide plating gold-plating imitation technology is more mature, imitating gold-tone stability, process maintenance, and is favored by enterprises. However, cyanide is highly toxic and has serious environmental pollution. Therefore, people turned to the development of non-cyanide pyrophosphate plating gold imitation process. Although its plating solution is low in toxicity and has little pollution to the environment, its stability is not good and it is difficult to control the imitating gold color. In addition, these two kinds of imitation gold electroplating process is long, all need to electroplating bright copper and bright nickel, equipment investment, higher production costs. Therefore, the development of a simple chemical imidization gold imitation process is imminent. The research group has used the method of direct electroless nickel plating and gold-plated chemical imitation plating to successfully develop a gold-plated chemical imitation gold-plating imitation gold process technology [3]. In order to further expand the application scope of the gold-plated chemical imitation gold plating process, the research group has also adopted a combination of secondary zinc immersion + alkaline electroless nickel plating + acidic electroless nickel plating + chemical leaching imitation gold to successfully develop a new one. Aluminum and aluminum alloy chemical imitation gold plating imitation gold technology, the formation of the imitation gold layer color is elegant and pure, can be comparable with the electroplated imitation gold layer, and the bonding force is good, the process is simple, has little pollution to the environment, and thus has a broad application prospects.
2. Experiment 2.1 Instruments and Drugs Experimental apparatus: HH-2 digital thermostatic water bath (Jintan Fuhua Instrument Co., Ltd.) and salt spray test box (Jiangsu Wuxi Sunan Test Equipment Co., Ltd.).
Chemicals: nickel sulphate, sodium hypophosphite, copper sulphate and stannous sulphate, etc.; industrial grade; sodium citrate, sodium acetate, sodium hydroxide, saccharin, hydrofluoric acid and ammonium fluoride, etc., commercially available in chemical purity.
Experimental aluminum: Industrial aluminum plate 6063, size 50mm × 60mm × (0.5 ~ 1.0) mm.
2.2 Performance Test 2.2.1 Coating Bonding Force Detection According to the bending test in GB/T 5270-2005 “Test Methods for Electrodeposition and Chemical Deposition of Metal Coatings on Metal Substratesâ€, the chemical dipping coating prepared by this process was simulated. The gold layer sample was repeatedly bent until it broke, and no peeling, chipping and delamination were deemed as qualified. At the same time, the sample of the chemical imitation gold plating prepared in this process was heated at 220° C. for 30 minutes using a thermal shock test. Then it was cooled in room temperature water, and no blistering, delamination, or delamination was considered to be acceptable.
2.2.2 Coating salt water resistance test To compare the corrosion resistance of the immersion plating layer in salt water, the electroplating imitation gold sheet and the chemical imitation gold plating imitation gold sheet of this process were simultaneously immersed in room temperature and 5% (mass fraction) NaCl solution. After a period of time, the corrosion of the surface of the test piece was observed, and no corrosion was observed in the test for 240 hours.
2.3 Process Flow Because of the active nature of aluminum chemistry, it has a strong affinity for oxygen. Therefore, whether gold and aluminum alloy chemical imitation gold plating is successful or not, the process combination is the key. Through the study and comparison, four kinds of combined technological processes were selected for research.
(1) Secondary Zinc Leaching + Alkaline Electroless Nickel Plating + Acidic Electroless Nickel Plating + Chemical Immersion Gold Imitation Gold Process: Alkaline Degreasing - Hot Water Washing - Flow Washing - Alkali Erosion - Flow Washing - Nitric Acid Light - Water Washing - Once Zinc leaching ─ Washing with water ─ 1:1 (volume ratio, the same below) Nitric acid dezincification ─ Washing with water ─ Secondary zinc leaching [4] ─ Washing with water ─ Deionized water washing ─ Alkaline electroless nickel plating [5] ─ Washing water ─ Acidic electroless nickel plating [6] - Flow washing - Imitation gold plating - Washing water - Passivation - Washing - Drying - Dipping varnish - Drying - Inspection.
(2) Secondary Zinc Leaching + Flash Nickel Plating + Acidic Electroless Nickel Plating + Chemical Immersion Gold Imitation Gold Process: Alkaline Degreasing - Hot Water Washing - Washing with Water - Alkali Erosion - Flow Washing - Nitric Acid Light - Water Washing - Zinc Dipping ( Zinc Oxide 100g/L, Sodium Hydroxide 500g/L, Potassium Sodium Tartrate 20g/L, Ferric chloride 1g/L, Temperature 15~25°C, Time 30~60s) - Washed with water ─ 1:1 Nitric Acid Dezincification - Washing with water - Secondary zinc leaching - Washing with water - Pre-plating with neutral nickel - Flow washing - Deionized water washing - Electroless nickel plating - Hot water washing - Hot water sealing (5 min) - Drying - Performance test.
(3) Pre-plated Nickel + Acidic Electroless Nickel Plating + Chemical Immersion Gold Imitation Gold Process: Alkaline Degreasing - Hot Washing - Flow Washing - Alkali Erosion - Flow Washing - Nitric Acid Light - Flow Washing - Pre-plating of Neutral Nickel - Electroless Plating Nickel-hot water washing-hot water sealing (5 min)-drying-performance test.
(4) Conditional Pretreatment + Pre-neutral Nickel + Acidic Electroless Nickel Plating + Chemical Immersion Plating Imitation Gold Process: Alkaline Degreasing - Hot Water Washing - Washing with Water - Alkali Erosion - Flow Washing - Nitric Acid Light - Water Washing - Surface Conditioning - Pre-nickel plating - Electroless nickel plating - Hot water washing - Hot water sealing (5 min) - Drying - Performance test.
3. Results and discussion 3.1 Determination of gold imitation gold plating process conditions The results of the four combinatorial process experiments are shown in Table 1. It can be seen that the combination process (1) can realize the imitation gold plating with chemical imitation, and the formed coating has a gold-like imitation, and the bonding force is good and the decorative effect is good.

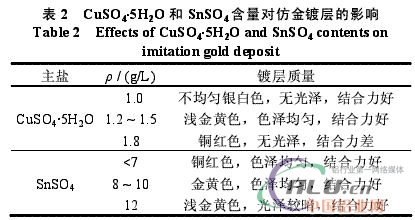
3.2 Effect and influence of main components 3.2.1 Effect of copper sulfate and stannous sulfate Copper sulfate and stannous sulfate are the main salts of imitation gold plating. In the acid environment of the plating bath, Cu2+ and Sn2+ form with the aluminum surface. The nickel layer undergoes a substitution reaction to form an imitation gold plating layer. By controlling the content of copper and tin in the coating, the iridescent gold can be obtained. The influence of the content of copper sulfate and stannous sulfate on the color of the imitating gold layer is shown in Table 2. According to Table 2, the copper sulfate content is controlled at 1.2~1.5g/L, and the SnSO4 content is controlled at 8~10g/L.
3.2.2 Influence of Coordination Agents and Accelerators Replacement of copper with metal nickel is much faster than displacing tin. Therefore, it is difficult to obtain an ideal gold-like layer in a simple mixed solution of Cu2+ and Sn2+, only when the displacement reaction of Cu2+ is affected. The gold imitation gold layer with a lower copper content can only be obtained if the limitation or the Sn2+ substitution reaction is properly promoted. Through experiments, tartaric acid or citric acid was selected as the copper ion complexing agent, and hydrofluoric acid was used as the tin ion accelerator to better adjust the replacement rate of Cu2+ and Sn2+ and obtain the ideal imitating gold color. The suitable content of the complexing agent is 10~15g/L, and the hydrofluoric acid content is 40~50mL/L.
3.2.3 Effect of Sulfuric Acid The role of sulfuric acid is to provide an acidic environment for the displacement reaction of copper and tin, as well as to inhibit the hydrolysis of Sn2+ in the bath to keep the bath stable.
The effect of sulfuric acid concentration on coating quality is shown in Table 3. As can be seen from Table 3, H2SO4 content is preferably controlled at 10-20 mL/L.

3.2.4 Effect of Stabilizer Because Sn2+ in the bath is very unstable, it is easily oxidized to Sn4+, and Sn4+ is easily hydrolyzed to produce precipitation, so that the effective content of Sn2+ in the plating solution is reduced, resulting in difficulty in replacement of tin, resulting in the copper color of the plating layer. color. In order to prevent or delay the oxidation of Sn2+ in the acidic bath and increase the stability of the bath, an effective stabilizer must be added. According to the literature [3] experience, combined with the characteristics of this process, a variety of organic compounds (hydroquinone, formaldehyde, ascorbic acid, citric acid) and inorganic compounds (strontium and vanadium compounds) were selected to prepare a highly effective compound stabilizer XT-08B The stabilizer has strong reducibility and can partially reduce Sn4+ to Sn2+. The chemical imitation gold plating solution without XT-08B stabilizer placed for 7 days will produce a large amount of precipitates; after adding the XT-08B stabilizer, the chemical imitation gold plating solution will remain for 3 months, still clarified, no precipitate, and its stability Greatly improved sex. In addition, the effect of XT-08B stabilizer on the color of the imitation gold coating is very small, and its addition amount is preferably controlled at 10-15 mL/L. Stabilizer content is too low, the stability effect is poor; too high, increase production costs.
3.3 Influence of process conditions When the temperature is increased, the relative content of relatively easily deposited metal copper in the alloy has a tendency to increase, resulting in increased copper content in the coating. In addition, the increase in temperature may lead to tin hydrolyzation, so that the content of SnSO4 in the bath decreases. General temperature control in 15 ~ 35 °C is appropriate. When the temperature is too high, the tin content in the coating is reduced, and the coating is copper-red, which affects the imitation gold color.
Plating time is controlled at 10~15min. If the time is too short, the imitated gold layer formed is light in color, uneven, and exposed at the end; over time, the imitation gold is darker and coppery red, which affects the decorative effect.
3.4 Performance Test Results 3.4.1 Measurement of Bonding Force In the bending test, the chemical imitation gold-plated imitation gold layer did not show peeling, chipping, and flaking; in the thermal shock test, the chemical imitation gold-plated imitation gold layer did not appear to foam. , peeling, cracks and other phenomena. This shows that the coating has a good bonding force.
3.4.2 Corrosion resistance to salt water To compare the corrosion resistance of the immersion plating layer in salt water, electroplating imitation gold flakes and the chemical imitation plating gold-imitation flakes of this process were simultaneously immersed in room temperature and 5% NaCl solution, and after 240 h immersion test, two kinds No corrosion of the test piece occurred. This shows that the imitation gold imitation gold layer formed by this process is equivalent to the electroplated imitation gold layer in corrosion resistance, and can substitute for gold imitation plating.
4. Conclusion A new method of imitation gold electroless plating for aluminum and aluminum alloys was developed using the combination of zinc immersion + alkaline electroless nickel plating + acidic electroless nickel plating + chemical immersion gold imitation gold plating process. Process specifications are as follows: SnSO48~10g/L, CuSO41.2~1.5g/L, coordination agent 10~15g/L, H2SO410~20mL/L, XT-08B stabilizer 10~12mL/L, hydrofluoric acid 40~ 50mL/L, ammonium fluoride 1~2g/L, temperature 15~35°C, time 10~15min. The imitation gold layer obtained by this process is elegant and pure in color, has good bonding force, is simple in process operation, and has less pollution to the environment, and the corrosion resistance can be comparable to the electroplated imitation gold layer, so it has a good application prospect.
Superabrasives come in the form of grinding wheels and are used when the material being processed is either too hard or too delicate for regular abrasives.
Industrial diamonds are one of the most common materials used to create the grinding material. Contrary to popular belief, diamonds are not as rare as marketing campaigns will tell you.
After diamonds,cubic boron nitride is the second more durable material for cutting. This is followed by polycrystalline.
The most obvious advantage is that these Grinding Wheels are extremely hard and durable. This allows them to give better performance and greater longevity over regular abrasives.
When your wheel lasts longer you will see reduced costs for fixtures and tooling. This opens up your budget up to investing in more machines.
You will notice that the wear and cuts on your materials will be even. Because the abrasiveness of the wheel is so strong, it doesn`t develop uneven worn away spots as quickly as regular abrasive wheels.
So the output from your production will be consistent. So your manufacturing efforts will produce reliable and consistent results.
It can be tough to justify the upfront increased costs of superabrasive wheels. But the durability and quality of their production output make them well worth it.
But you`ll quickly realize that your productivity has increased by using superabrasives. You will have more parts processed per wheel and fewer wheel changes.
Kemei produces both conventional grinding wheel and super abrasive grinding wheels, such as white aluminum grinding wheel, pink wheel ,green silicon wheel, diamond grinding wheel and CBN grinding wheels.
If you need any type of grinding wheel and any suggestion on grinding wheels, just contact us.
Super Abrasives,Grinding Wheel Stone,Grinding Wheel For Bench Grinder,Electroplated Cbn Cut Off Wheel
Henan Jinlun Superhard Material Co., Ltd , https://www.kmgrindingwheels.com